Capping A (Tetrahydrofuran 26-Lutidine Acetic Anhydride V V V 80 10 10) PurOligo
PurOligo
Properties
| Vapour pressure | No data available |
| Water Solubility | Soluble |
| Autoignition temp. | No data available |
| Explosion limit | Upper explosion limit: 12.4 %(V) Lower explosion limit: 1.5 %(V) |
| Refractive index | 1.412 â 1.416 |
| B pt. | No data available |
| M pt. | Nodataavailable |
| Density | No data available |
| Form | Liquid |
Safety Information
| Hazard Statement(s) | H225-H302+H332-H315-H318-H335-H351-EUH019-EUH071 |
| Precautionary Statement | P280-P301+P312-P304+P340-P312-P305+P351+P338-P308+P313 |
| GHS Pictogram |
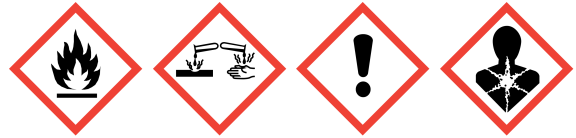
|
| Signal word | Danger |
| Flash point | -20°C |
| Storage Temp. | 2-30°C |
| Storage Class | 3 - Flammable liquids |
| UN Number | 2924 |
Description
Other Notes
For each synthesis cycle up to 1 to 2 % of free 5-hydroxy groups remain after the phosphoramidite coupling step has been completed. By running a subsequent Capping step using an anhydride these free hydroxyl groups are converted to acetates and are hindered from further chain elongation and formation of long oligonucleotides with incorrect sequences. For optimal acetylation a solution of acetic anhydride in Tetrahydrofuran or acetonitrile (Capping A) will be mixed in situ during reaction with a catalytic acting solution of N-methylimidazole (Capping B). Additives such as pyridine and lutidine function as mild bases to enhance the efficiency of the capping reaction.
Application
Capping Reagent for Nucleic Acid Synthesis.
Purpose
Laboratory use
Documents
| SDS | |
| COA | |
| Specification | |
| Bulk quote order form |
